Copper »
PDB 6pvz-6ri0 »
6rhh »
Copper in PDB 6rhh: Single Crystal Serial Study of the X-Ray Induced Enzymatic Reduction of Molecular Oxygen to Water For Laccase From Steccherinum Murashkinskyi at Sub-Atomic Resolution. Third Structure of the Series with 315 Kgy Dose.
Enzymatic activity of Single Crystal Serial Study of the X-Ray Induced Enzymatic Reduction of Molecular Oxygen to Water For Laccase From Steccherinum Murashkinskyi at Sub-Atomic Resolution. Third Structure of the Series with 315 Kgy Dose.
All present enzymatic activity of Single Crystal Serial Study of the X-Ray Induced Enzymatic Reduction of Molecular Oxygen to Water For Laccase From Steccherinum Murashkinskyi at Sub-Atomic Resolution. Third Structure of the Series with 315 Kgy Dose.:
1.10.3.2;
1.10.3.2;
Protein crystallography data
The structure of Single Crystal Serial Study of the X-Ray Induced Enzymatic Reduction of Molecular Oxygen to Water For Laccase From Steccherinum Murashkinskyi at Sub-Atomic Resolution. Third Structure of the Series with 315 Kgy Dose., PDB code: 6rhh
was solved by
K.M.Polyakov,
S.Gavryushov,
T.V.Fedorova,
O.A.Glazunova,
A.N.Popov,
with X-Ray Crystallography technique. A brief refinement statistics is given in the table below:
| Resolution Low / High (Å) | 67.34 / 0.97 |
| Space group | P 21 21 21 |
| Cell size a, b, c (Å), α, β, γ (°) | 56.250, 84.130, 112.360, 90.00, 90.00, 90.00 |
| R / Rfree (%) | 11.5 / 13.1 |
Copper Binding Sites:
The binding sites of Copper atom in the Single Crystal Serial Study of the X-Ray Induced Enzymatic Reduction of Molecular Oxygen to Water For Laccase From Steccherinum Murashkinskyi at Sub-Atomic Resolution. Third Structure of the Series with 315 Kgy Dose.
(pdb code 6rhh). This binding sites where shown within
5.0 Angstroms radius around Copper atom.
In total 7 binding sites of Copper where determined in the Single Crystal Serial Study of the X-Ray Induced Enzymatic Reduction of Molecular Oxygen to Water For Laccase From Steccherinum Murashkinskyi at Sub-Atomic Resolution. Third Structure of the Series with 315 Kgy Dose., PDB code: 6rhh:
Jump to Copper binding site number: 1; 2; 3; 4; 5; 6; 7;
In total 7 binding sites of Copper where determined in the Single Crystal Serial Study of the X-Ray Induced Enzymatic Reduction of Molecular Oxygen to Water For Laccase From Steccherinum Murashkinskyi at Sub-Atomic Resolution. Third Structure of the Series with 315 Kgy Dose., PDB code: 6rhh:
Jump to Copper binding site number: 1; 2; 3; 4; 5; 6; 7;
Copper binding site 1 out of 7 in 6rhh
Go back to
Copper binding site 1 out
of 7 in the Single Crystal Serial Study of the X-Ray Induced Enzymatic Reduction of Molecular Oxygen to Water For Laccase From Steccherinum Murashkinskyi at Sub-Atomic Resolution. Third Structure of the Series with 315 Kgy Dose.
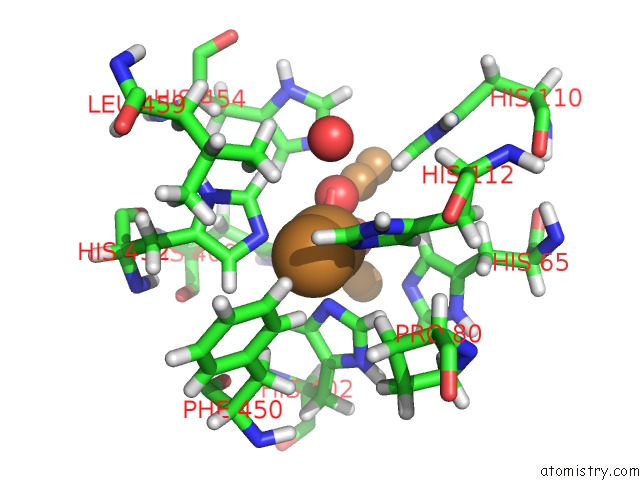
Mono view
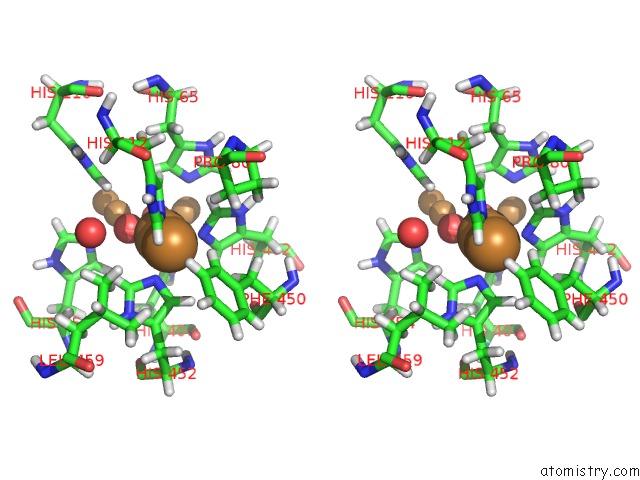
Stereo pair view

Mono view

Stereo pair view
A full contact list of Copper with other atoms in the Cu binding
site number 1 of Single Crystal Serial Study of the X-Ray Induced Enzymatic Reduction of Molecular Oxygen to Water For Laccase From Steccherinum Murashkinskyi at Sub-Atomic Resolution. Third Structure of the Series with 315 Kgy Dose. within 5.0Å range:
|
Copper binding site 2 out of 7 in 6rhh
Go back to
Copper binding site 2 out
of 7 in the Single Crystal Serial Study of the X-Ray Induced Enzymatic Reduction of Molecular Oxygen to Water For Laccase From Steccherinum Murashkinskyi at Sub-Atomic Resolution. Third Structure of the Series with 315 Kgy Dose.
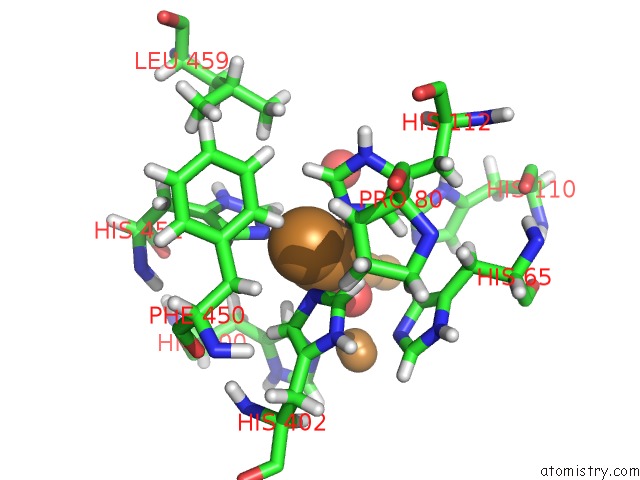
Mono view
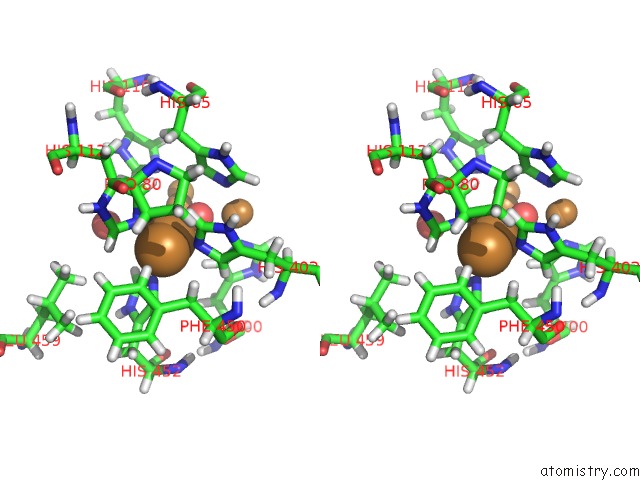
Stereo pair view

Mono view

Stereo pair view
A full contact list of Copper with other atoms in the Cu binding
site number 2 of Single Crystal Serial Study of the X-Ray Induced Enzymatic Reduction of Molecular Oxygen to Water For Laccase From Steccherinum Murashkinskyi at Sub-Atomic Resolution. Third Structure of the Series with 315 Kgy Dose. within 5.0Å range:
|
Copper binding site 3 out of 7 in 6rhh
Go back to
Copper binding site 3 out
of 7 in the Single Crystal Serial Study of the X-Ray Induced Enzymatic Reduction of Molecular Oxygen to Water For Laccase From Steccherinum Murashkinskyi at Sub-Atomic Resolution. Third Structure of the Series with 315 Kgy Dose.
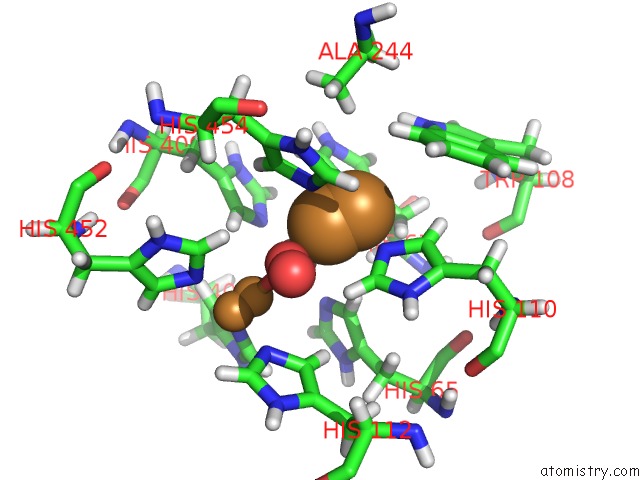
Mono view
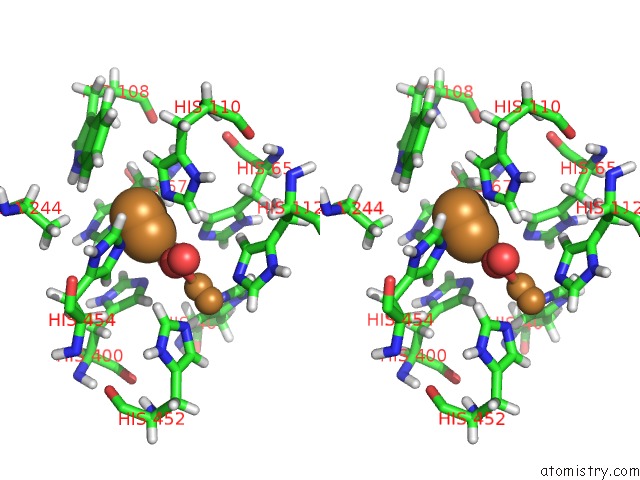
Stereo pair view

Mono view

Stereo pair view
A full contact list of Copper with other atoms in the Cu binding
site number 3 of Single Crystal Serial Study of the X-Ray Induced Enzymatic Reduction of Molecular Oxygen to Water For Laccase From Steccherinum Murashkinskyi at Sub-Atomic Resolution. Third Structure of the Series with 315 Kgy Dose. within 5.0Å range:
|
Copper binding site 4 out of 7 in 6rhh
Go back to
Copper binding site 4 out
of 7 in the Single Crystal Serial Study of the X-Ray Induced Enzymatic Reduction of Molecular Oxygen to Water For Laccase From Steccherinum Murashkinskyi at Sub-Atomic Resolution. Third Structure of the Series with 315 Kgy Dose.
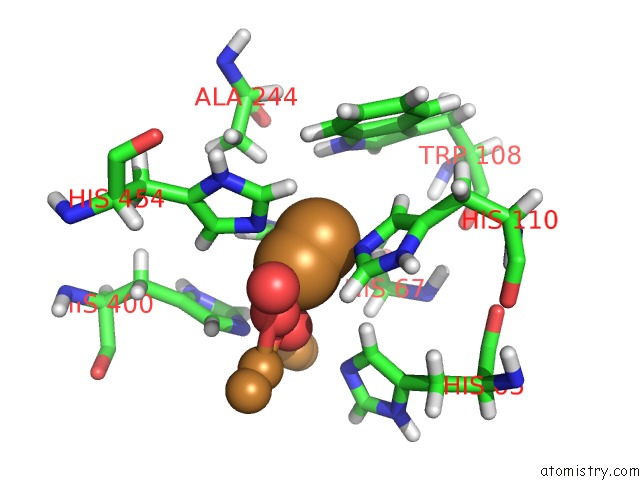
Mono view
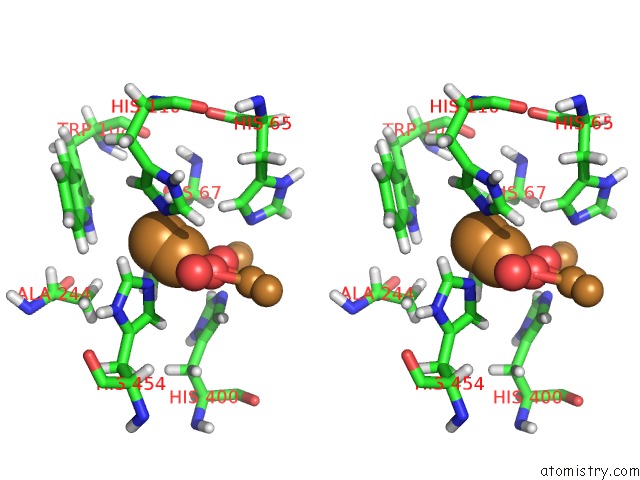
Stereo pair view

Mono view

Stereo pair view
A full contact list of Copper with other atoms in the Cu binding
site number 4 of Single Crystal Serial Study of the X-Ray Induced Enzymatic Reduction of Molecular Oxygen to Water For Laccase From Steccherinum Murashkinskyi at Sub-Atomic Resolution. Third Structure of the Series with 315 Kgy Dose. within 5.0Å range:
|
Copper binding site 5 out of 7 in 6rhh
Go back to
Copper binding site 5 out
of 7 in the Single Crystal Serial Study of the X-Ray Induced Enzymatic Reduction of Molecular Oxygen to Water For Laccase From Steccherinum Murashkinskyi at Sub-Atomic Resolution. Third Structure of the Series with 315 Kgy Dose.
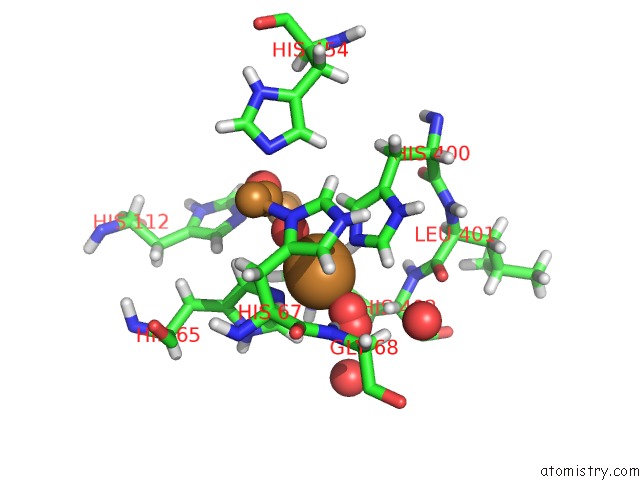
Mono view
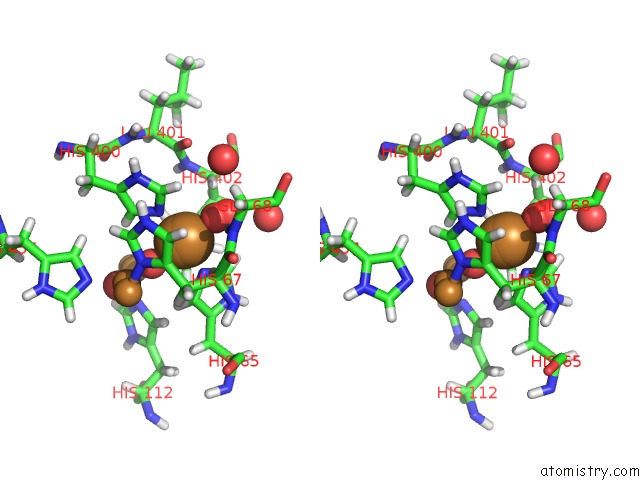
Stereo pair view

Mono view

Stereo pair view
A full contact list of Copper with other atoms in the Cu binding
site number 5 of Single Crystal Serial Study of the X-Ray Induced Enzymatic Reduction of Molecular Oxygen to Water For Laccase From Steccherinum Murashkinskyi at Sub-Atomic Resolution. Third Structure of the Series with 315 Kgy Dose. within 5.0Å range:
|
Copper binding site 6 out of 7 in 6rhh
Go back to
Copper binding site 6 out
of 7 in the Single Crystal Serial Study of the X-Ray Induced Enzymatic Reduction of Molecular Oxygen to Water For Laccase From Steccherinum Murashkinskyi at Sub-Atomic Resolution. Third Structure of the Series with 315 Kgy Dose.
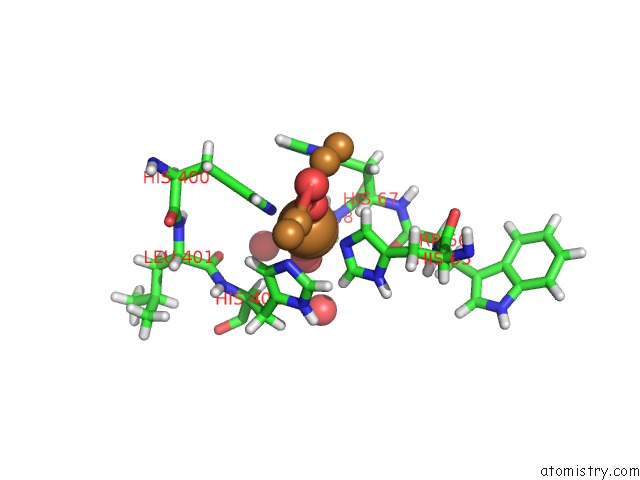
Mono view
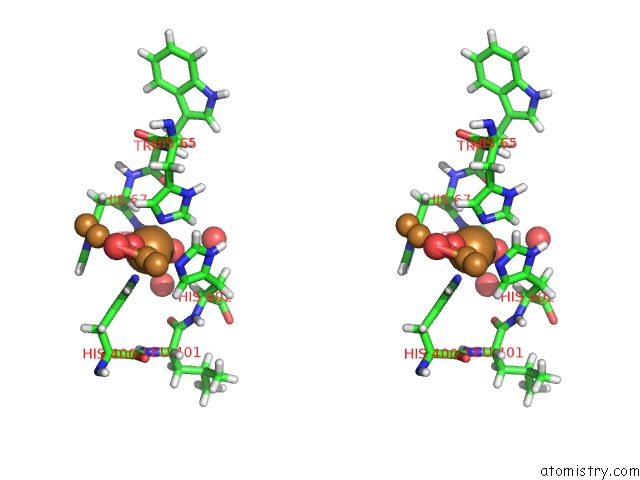
Stereo pair view

Mono view

Stereo pair view
A full contact list of Copper with other atoms in the Cu binding
site number 6 of Single Crystal Serial Study of the X-Ray Induced Enzymatic Reduction of Molecular Oxygen to Water For Laccase From Steccherinum Murashkinskyi at Sub-Atomic Resolution. Third Structure of the Series with 315 Kgy Dose. within 5.0Å range:
|
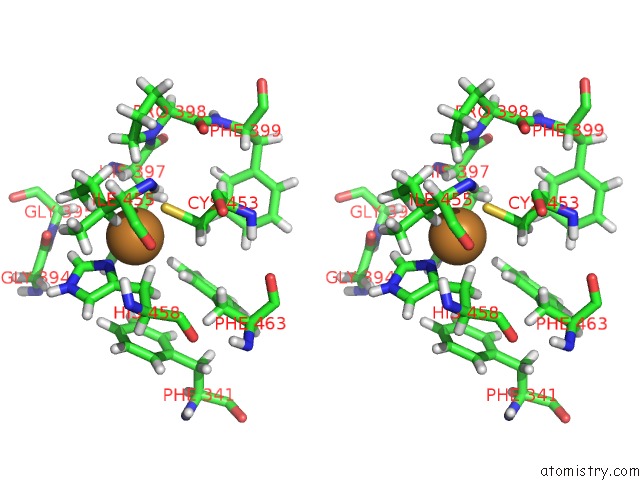
Copper binding site 7 out of 7 in 6rhh
Go back to
Copper binding site 7 out
of 7 in the Single Crystal Serial Study of the X-Ray Induced Enzymatic Reduction of Molecular Oxygen to Water For Laccase From Steccherinum Murashkinskyi at Sub-Atomic Resolution. Third Structure of the Series with 315 Kgy Dose.
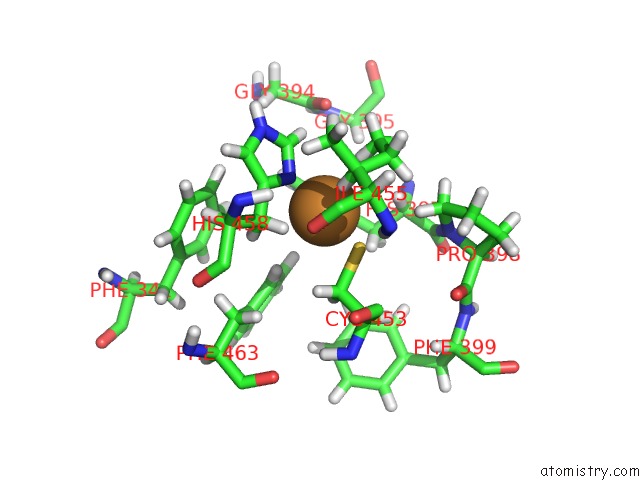
Mono view
Stereo pair view

Mono view

Stereo pair view
A full contact list of Copper with other atoms in the Cu binding
site number 7 of Single Crystal Serial Study of the X-Ray Induced Enzymatic Reduction of Molecular Oxygen to Water For Laccase From Steccherinum Murashkinskyi at Sub-Atomic Resolution. Third Structure of the Series with 315 Kgy Dose. within 5.0Å range:
|
Reference:
K.M.Polyakov,
S.Gavryushov,
T.V.Fedorova,
O.A.Glazunova,
A.N.Popov.
The Subatomic Resolution Study of Laccase Inhibition By Chloride and Fluoride Anions Using Single-Crystal Serial Crystallography: Insights Into the Enzymatic Reaction Mechanism. Acta Crystallogr D Struct V. 75 804 2019BIOL.
ISSN: ISSN 2059-7983
PubMed: 31478903
DOI: 10.1107/S2059798319010684
Page generated: Wed Jul 31 06:59:43 2024
ISSN: ISSN 2059-7983
PubMed: 31478903
DOI: 10.1107/S2059798319010684
Last articles
Zn in 9J0NZn in 9J0O
Zn in 9J0P
Zn in 9FJX
Zn in 9EKB
Zn in 9C0F
Zn in 9CAH
Zn in 9CH0
Zn in 9CH3
Zn in 9CH1