Copper »
PDB 6fok-6ibj »
6gsq »
Copper in PDB 6gsq: Oxidised Copper Nitrite Reductase From Achromobacter Cycloclastes Determined By Serial Femtosecond Rotation Crystallography
Enzymatic activity of Oxidised Copper Nitrite Reductase From Achromobacter Cycloclastes Determined By Serial Femtosecond Rotation Crystallography
All present enzymatic activity of Oxidised Copper Nitrite Reductase From Achromobacter Cycloclastes Determined By Serial Femtosecond Rotation Crystallography:
1.7.2.1;
1.7.2.1;
Protein crystallography data
The structure of Oxidised Copper Nitrite Reductase From Achromobacter Cycloclastes Determined By Serial Femtosecond Rotation Crystallography, PDB code: 6gsq
was solved by
T.P.Halsted,
K.Yamashita,
C.C.Gopalasingam,
R.T.Shenoy,
K.Hirata,
H.Ago,
G.Ueno,
R.R.Eady,
S.V.Antonyuk,
M.Yamamoto,
S.S.Hasnain,
with X-Ray Crystallography technique. A brief refinement statistics is given in the table below:
| Resolution Low / High (Å) | 54.88 / 1.50 |
| Space group | P 21 3 |
| Cell size a, b, c (Å), α, β, γ (°) | 94.950, 94.950, 94.950, 90.00, 90.00, 90.00 |
| R / Rfree (%) | 14.4 / 17.7 |
Copper Binding Sites:
The binding sites of Copper atom in the Oxidised Copper Nitrite Reductase From Achromobacter Cycloclastes Determined By Serial Femtosecond Rotation Crystallography
(pdb code 6gsq). This binding sites where shown within
5.0 Angstroms radius around Copper atom.
In total 2 binding sites of Copper where determined in the Oxidised Copper Nitrite Reductase From Achromobacter Cycloclastes Determined By Serial Femtosecond Rotation Crystallography, PDB code: 6gsq:
Jump to Copper binding site number: 1; 2;
In total 2 binding sites of Copper where determined in the Oxidised Copper Nitrite Reductase From Achromobacter Cycloclastes Determined By Serial Femtosecond Rotation Crystallography, PDB code: 6gsq:
Jump to Copper binding site number: 1; 2;
Copper binding site 1 out of 2 in 6gsq
Go back to
Copper binding site 1 out
of 2 in the Oxidised Copper Nitrite Reductase From Achromobacter Cycloclastes Determined By Serial Femtosecond Rotation Crystallography
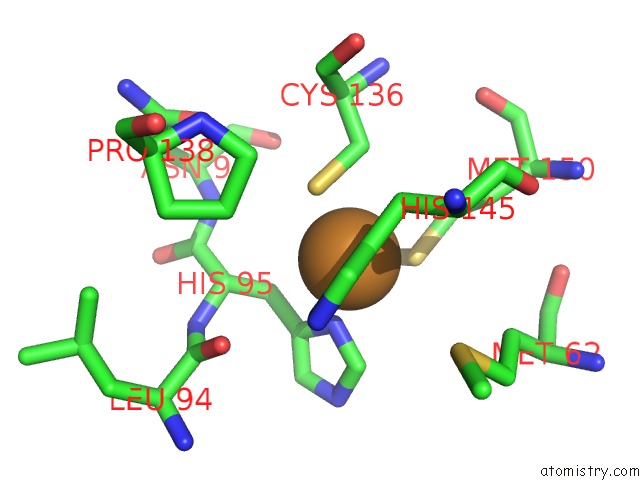
Mono view

Stereo pair view

Mono view

Stereo pair view
A full contact list of Copper with other atoms in the Cu binding
site number 1 of Oxidised Copper Nitrite Reductase From Achromobacter Cycloclastes Determined By Serial Femtosecond Rotation Crystallography within 5.0Å range:
|
Copper binding site 2 out of 2 in 6gsq
Go back to
Copper binding site 2 out
of 2 in the Oxidised Copper Nitrite Reductase From Achromobacter Cycloclastes Determined By Serial Femtosecond Rotation Crystallography

Mono view
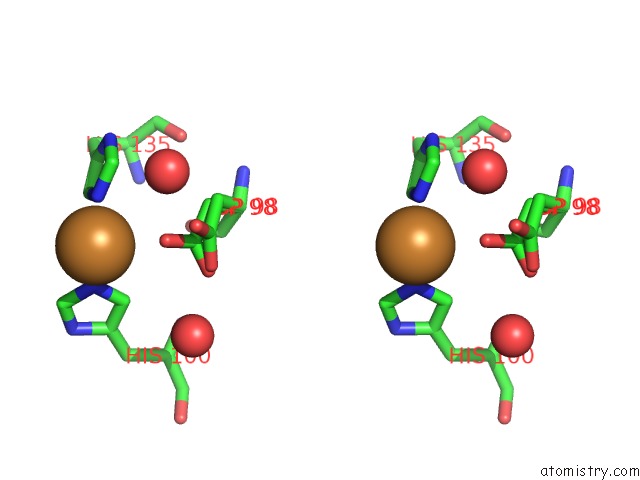
Stereo pair view

Mono view

Stereo pair view
A full contact list of Copper with other atoms in the Cu binding
site number 2 of Oxidised Copper Nitrite Reductase From Achromobacter Cycloclastes Determined By Serial Femtosecond Rotation Crystallography within 5.0Å range:
|
Reference:
T.P.Halsted,
K.Yamashita,
C.C.Gopalasingam,
R.T.Shenoy,
K.Hirata,
H.Ago,
G.Ueno,
M.P.Blakeley,
R.R.Eady,
S.V.Antonyuk,
M.Yamamoto,
S.S.Hasnain.
Catalytically Important Damage-Free Structures of A Copper Nitrite Reductase Obtained By Femtosecond X-Ray Laser and Room-Temperature Neutron Crystallography. Iucrj V. 6 761 2019.
ISSN: ESSN 2052-2525
PubMed: 31316819
DOI: 10.1107/S2052252519008285
Page generated: Mon Jul 14 06:09:21 2025
ISSN: ESSN 2052-2525
PubMed: 31316819
DOI: 10.1107/S2052252519008285
Last articles
F in 4IFVF in 4IDO
F in 4ICC
F in 4IBI
F in 4IAH
F in 4IAE
F in 4I9H
F in 4I9N
F in 4I9O
F in 4IA9