Copper »
PDB 3iea-3mn0 »
3l45 »
Copper in PDB 3l45: A Joint Neutron and X-Ray Structure of Oxidized Amicyanin
Protein crystallography data
The structure of A Joint Neutron and X-Ray Structure of Oxidized Amicyanin, PDB code: 3l45
was solved by
N.Sukumar,
F.S.Mathews,
P.Langan,
V.L.Davidson,
with X-Ray Crystallography technique. A brief refinement statistics is given in the table below:
| Resolution Low / High (Å) | N/A / 1.80 |
| Space group | P 1 21 1 |
| Cell size a, b, c (Å), α, β, γ (°) | 27.540, 56.580, 28.860, 90.00, 96.21, 90.00 |
| R / Rfree (%) | 19.8 / 21.5 |
Copper Binding Sites:
The binding sites of Copper atom in the A Joint Neutron and X-Ray Structure of Oxidized Amicyanin
(pdb code 3l45). This binding sites where shown within
5.0 Angstroms radius around Copper atom.
In total only one binding site of Copper was determined in the A Joint Neutron and X-Ray Structure of Oxidized Amicyanin, PDB code: 3l45:
In total only one binding site of Copper was determined in the A Joint Neutron and X-Ray Structure of Oxidized Amicyanin, PDB code: 3l45:
Copper binding site 1 out of 1 in 3l45
Go back to
Copper binding site 1 out
of 1 in the A Joint Neutron and X-Ray Structure of Oxidized Amicyanin
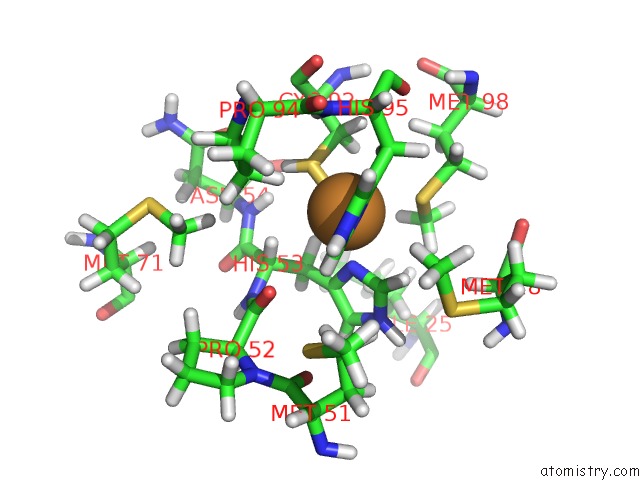
Mono view
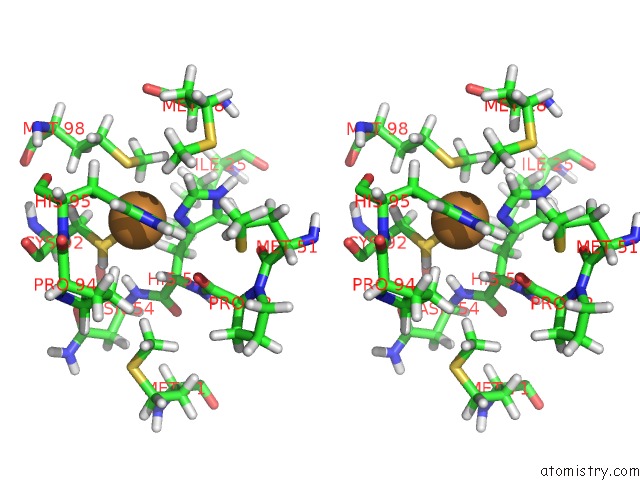
Stereo pair view

Mono view

Stereo pair view
A full contact list of Copper with other atoms in the Cu binding
site number 1 of A Joint Neutron and X-Ray Structure of Oxidized Amicyanin within 5.0Å range:
|
Reference:
N.Sukumar,
F.S.Mathews,
P.Langan,
V.L.Davidson.
A Joint X-Ray and Neutron Study on Amicyanin Reveals the Role of Protein Dynamics in Electron Transfer. Proc.Natl.Acad.Sci.Usa V. 107 6817 2010.
ISSN: ISSN 0027-8424
PubMed: 20351252
DOI: 10.1073/PNAS.0912672107
Page generated: Wed Jul 31 01:15:51 2024
ISSN: ISSN 0027-8424
PubMed: 20351252
DOI: 10.1073/PNAS.0912672107
Last articles
Cl in 3G4RCl in 3G4Q
Cl in 3G4L
Cl in 3G3K
Cl in 3G3X
Cl in 3G49
Cl in 3G3V
Cl in 3G3W
Cl in 3G3J
Cl in 3G3G