Copper »
PDB 2z7y-3aws »
2zxw »
Copper in PDB 2zxw: Bovine Heart Cytochrome C Oxidase at the Fully Oxidized State (1-S X- Ray Exposure Dataset)
Enzymatic activity of Bovine Heart Cytochrome C Oxidase at the Fully Oxidized State (1-S X- Ray Exposure Dataset)
All present enzymatic activity of Bovine Heart Cytochrome C Oxidase at the Fully Oxidized State (1-S X- Ray Exposure Dataset):
1.9.3.1;
1.9.3.1;
Protein crystallography data
The structure of Bovine Heart Cytochrome C Oxidase at the Fully Oxidized State (1-S X- Ray Exposure Dataset), PDB code: 2zxw
was solved by
H.Aoyama,
K.Muramoto,
K.Shinzawa-Itoh,
K.Hirata,
E.Yamashita,
T.Tsukihara,
T.Ogura,
S.Yoshikawa,
with X-Ray Crystallography technique. A brief refinement statistics is given in the table below:
| Resolution Low / High (Å) | 40.00 / 2.50 |
| Space group | P 21 21 21 |
| Cell size a, b, c (Å), α, β, γ (°) | 184.156, 207.621, 178.247, 90.00, 90.00, 90.00 |
| R / Rfree (%) | 18.5 / 23.3 |
Other elements in 2zxw:
The structure of Bovine Heart Cytochrome C Oxidase at the Fully Oxidized State (1-S X- Ray Exposure Dataset) also contains other interesting chemical elements:
| Magnesium | (Mg) | 2 atoms |
| Zinc | (Zn) | 2 atoms |
| Iron | (Fe) | 4 atoms |
| Sodium | (Na) | 2 atoms |
Copper Binding Sites:
The binding sites of Copper atom in the Bovine Heart Cytochrome C Oxidase at the Fully Oxidized State (1-S X- Ray Exposure Dataset)
(pdb code 2zxw). This binding sites where shown within
5.0 Angstroms radius around Copper atom.
In total 6 binding sites of Copper where determined in the Bovine Heart Cytochrome C Oxidase at the Fully Oxidized State (1-S X- Ray Exposure Dataset), PDB code: 2zxw:
Jump to Copper binding site number: 1; 2; 3; 4; 5; 6;
In total 6 binding sites of Copper where determined in the Bovine Heart Cytochrome C Oxidase at the Fully Oxidized State (1-S X- Ray Exposure Dataset), PDB code: 2zxw:
Jump to Copper binding site number: 1; 2; 3; 4; 5; 6;
Copper binding site 1 out of 6 in 2zxw
Go back to
Copper binding site 1 out
of 6 in the Bovine Heart Cytochrome C Oxidase at the Fully Oxidized State (1-S X- Ray Exposure Dataset)

Mono view

Stereo pair view

Mono view

Stereo pair view
A full contact list of Copper with other atoms in the Cu binding
site number 1 of Bovine Heart Cytochrome C Oxidase at the Fully Oxidized State (1-S X- Ray Exposure Dataset) within 5.0Å range:
|
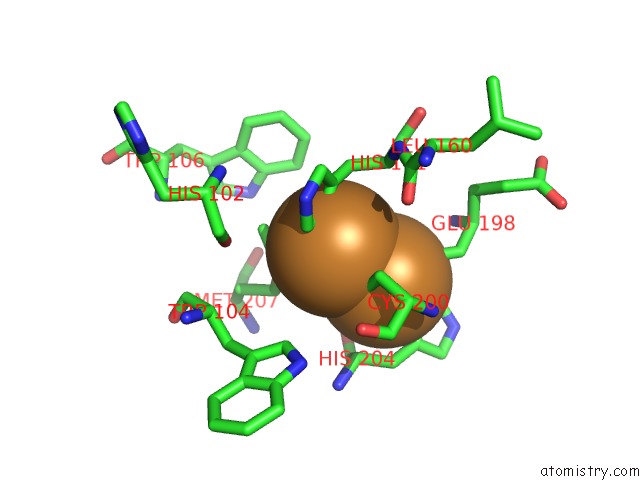
Copper binding site 2 out of 6 in 2zxw
Go back to
Copper binding site 2 out
of 6 in the Bovine Heart Cytochrome C Oxidase at the Fully Oxidized State (1-S X- Ray Exposure Dataset)
Mono view

Stereo pair view

Mono view

Stereo pair view
A full contact list of Copper with other atoms in the Cu binding
site number 2 of Bovine Heart Cytochrome C Oxidase at the Fully Oxidized State (1-S X- Ray Exposure Dataset) within 5.0Å range:
|
Copper binding site 3 out of 6 in 2zxw
Go back to
Copper binding site 3 out
of 6 in the Bovine Heart Cytochrome C Oxidase at the Fully Oxidized State (1-S X- Ray Exposure Dataset)

Mono view

Stereo pair view

Mono view

Stereo pair view
A full contact list of Copper with other atoms in the Cu binding
site number 3 of Bovine Heart Cytochrome C Oxidase at the Fully Oxidized State (1-S X- Ray Exposure Dataset) within 5.0Å range:
|
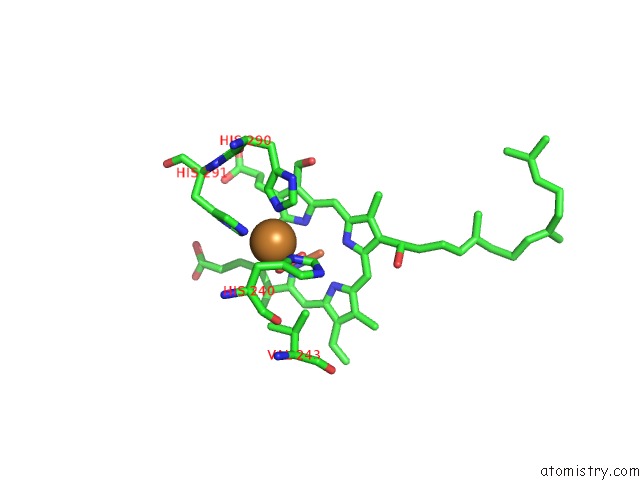
Copper binding site 4 out of 6 in 2zxw
Go back to
Copper binding site 4 out
of 6 in the Bovine Heart Cytochrome C Oxidase at the Fully Oxidized State (1-S X- Ray Exposure Dataset)
Mono view

Stereo pair view

Mono view

Stereo pair view
A full contact list of Copper with other atoms in the Cu binding
site number 4 of Bovine Heart Cytochrome C Oxidase at the Fully Oxidized State (1-S X- Ray Exposure Dataset) within 5.0Å range:
|
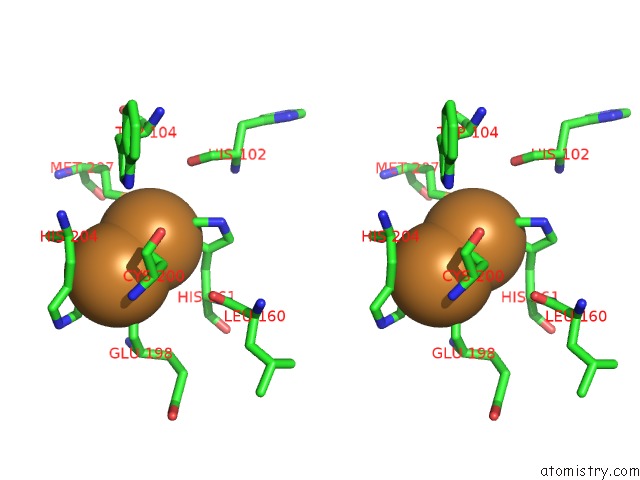
Copper binding site 5 out of 6 in 2zxw
Go back to
Copper binding site 5 out
of 6 in the Bovine Heart Cytochrome C Oxidase at the Fully Oxidized State (1-S X- Ray Exposure Dataset)

Mono view
Stereo pair view

Mono view

Stereo pair view
A full contact list of Copper with other atoms in the Cu binding
site number 5 of Bovine Heart Cytochrome C Oxidase at the Fully Oxidized State (1-S X- Ray Exposure Dataset) within 5.0Å range:
|
Copper binding site 6 out of 6 in 2zxw
Go back to
Copper binding site 6 out
of 6 in the Bovine Heart Cytochrome C Oxidase at the Fully Oxidized State (1-S X- Ray Exposure Dataset)

Mono view

Stereo pair view

Mono view

Stereo pair view
A full contact list of Copper with other atoms in the Cu binding
site number 6 of Bovine Heart Cytochrome C Oxidase at the Fully Oxidized State (1-S X- Ray Exposure Dataset) within 5.0Å range:
|
Reference:
H.Aoyama,
K.Muramoto,
K.Shinzawa-Itoh,
K.Hirata,
E.Yamashita,
T.Tsukihara,
T.Ogura,
S.Yoshikawa.
A Peroxide Bridge Between Fe and Cu Ions in the O2 Reduction Site of Fully Oxidized Cytochrome C Oxidase Could Suppress the Proton Pump Proc.Natl.Acad.Sci.Usa V. 106 2165 2009.
ISSN: ISSN 0027-8424
PubMed: 19164527
DOI: 10.1073/PNAS.0806391106
Page generated: Mon Jul 14 01:49:33 2025
ISSN: ISSN 0027-8424
PubMed: 19164527
DOI: 10.1073/PNAS.0806391106
Last articles
F in 7Q2YF in 7Q3B
F in 7Q2X
F in 7PVK
F in 7Q2J
F in 7Q01
F in 7PZX
F in 7PZW
F in 7PZV
F in 7PZU