Copper »
PDB 2cj3-2foy »
2fjs »
Copper in PDB 2fjs: Crystal Structure of Anaerobically Reduced Wild Type Nitrite Reductase From A. Faecalis
Enzymatic activity of Crystal Structure of Anaerobically Reduced Wild Type Nitrite Reductase From A. Faecalis
All present enzymatic activity of Crystal Structure of Anaerobically Reduced Wild Type Nitrite Reductase From A. Faecalis:
1.7.2.1;
1.7.2.1;
Protein crystallography data
The structure of Crystal Structure of Anaerobically Reduced Wild Type Nitrite Reductase From A. Faecalis, PDB code: 2fjs
was solved by
E.I.Tocheva,
M.E.P.Murphy,
with X-Ray Crystallography technique. A brief refinement statistics is given in the table below:
| Resolution Low / High (Å) | 30.00 / 1.85 |
| Space group | P 21 21 21 |
| Cell size a, b, c (Å), α, β, γ (°) | 61.552, 102.428, 145.744, 90.00, 90.00, 90.00 |
| R / Rfree (%) | 15 / 18 |
Copper Binding Sites:
The binding sites of Copper atom in the Crystal Structure of Anaerobically Reduced Wild Type Nitrite Reductase From A. Faecalis
(pdb code 2fjs). This binding sites where shown within
5.0 Angstroms radius around Copper atom.
In total 6 binding sites of Copper where determined in the Crystal Structure of Anaerobically Reduced Wild Type Nitrite Reductase From A. Faecalis, PDB code: 2fjs:
Jump to Copper binding site number: 1; 2; 3; 4; 5; 6;
In total 6 binding sites of Copper where determined in the Crystal Structure of Anaerobically Reduced Wild Type Nitrite Reductase From A. Faecalis, PDB code: 2fjs:
Jump to Copper binding site number: 1; 2; 3; 4; 5; 6;
Copper binding site 1 out of 6 in 2fjs
Go back to
Copper binding site 1 out
of 6 in the Crystal Structure of Anaerobically Reduced Wild Type Nitrite Reductase From A. Faecalis
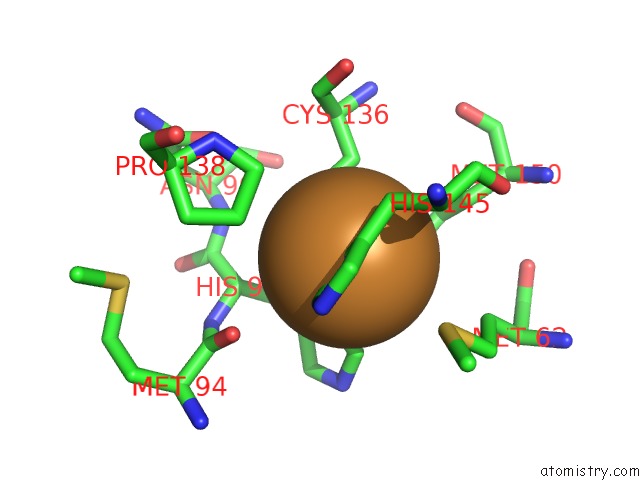
Mono view
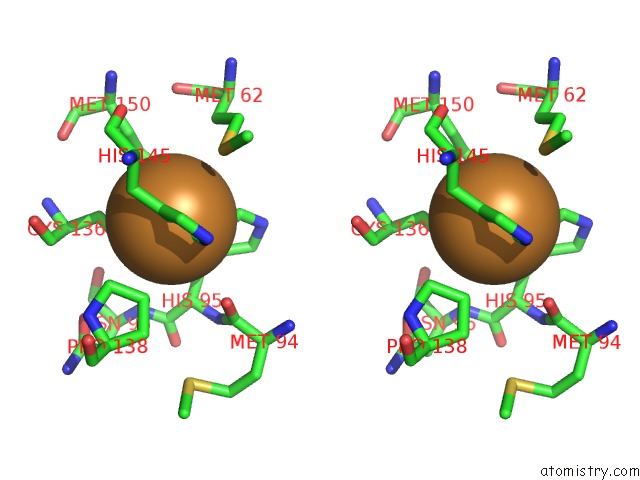
Stereo pair view

Mono view

Stereo pair view
A full contact list of Copper with other atoms in the Cu binding
site number 1 of Crystal Structure of Anaerobically Reduced Wild Type Nitrite Reductase From A. Faecalis within 5.0Å range:
|
Copper binding site 2 out of 6 in 2fjs
Go back to
Copper binding site 2 out
of 6 in the Crystal Structure of Anaerobically Reduced Wild Type Nitrite Reductase From A. Faecalis
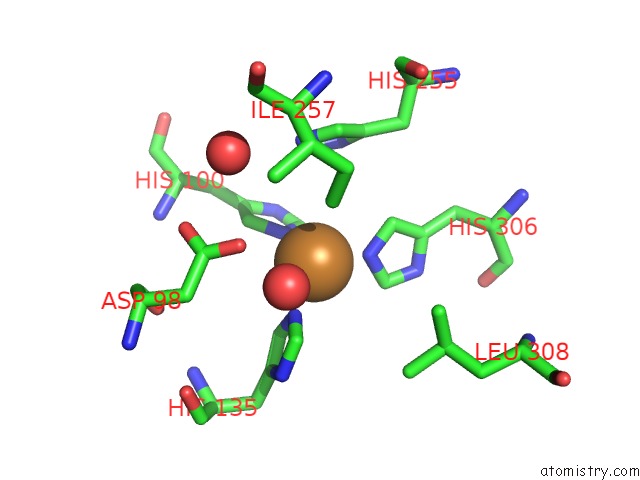
Mono view
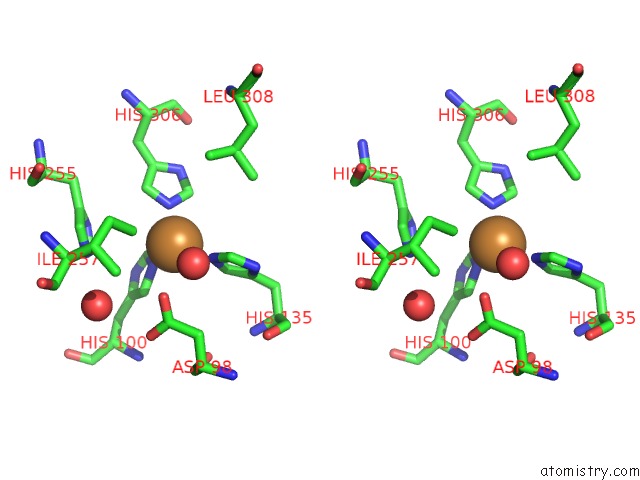
Stereo pair view

Mono view

Stereo pair view
A full contact list of Copper with other atoms in the Cu binding
site number 2 of Crystal Structure of Anaerobically Reduced Wild Type Nitrite Reductase From A. Faecalis within 5.0Å range:
|
Copper binding site 3 out of 6 in 2fjs
Go back to
Copper binding site 3 out
of 6 in the Crystal Structure of Anaerobically Reduced Wild Type Nitrite Reductase From A. Faecalis
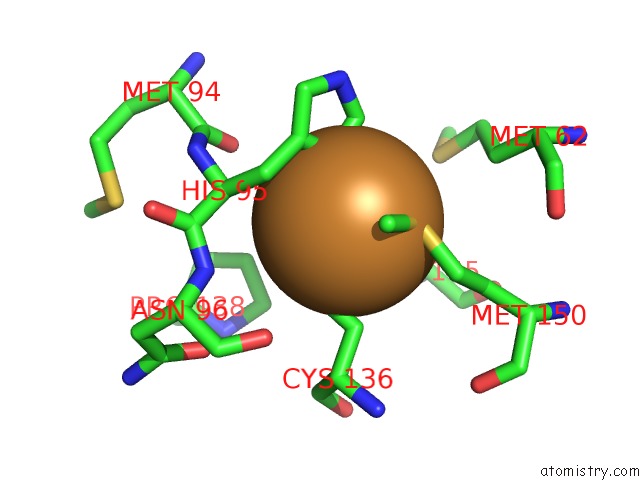
Mono view
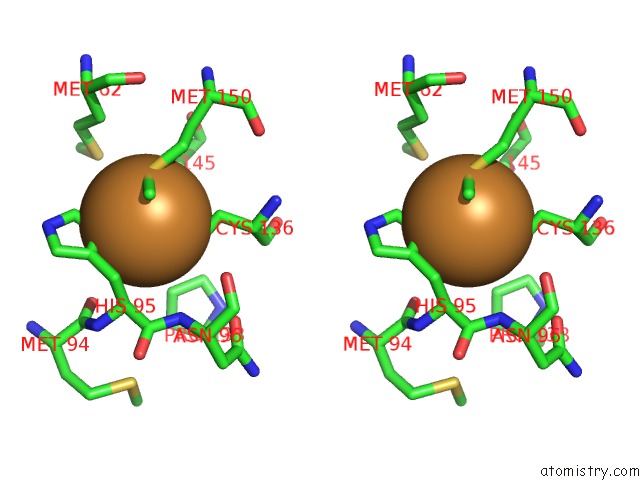
Stereo pair view

Mono view

Stereo pair view
A full contact list of Copper with other atoms in the Cu binding
site number 3 of Crystal Structure of Anaerobically Reduced Wild Type Nitrite Reductase From A. Faecalis within 5.0Å range:
|
Copper binding site 4 out of 6 in 2fjs
Go back to
Copper binding site 4 out
of 6 in the Crystal Structure of Anaerobically Reduced Wild Type Nitrite Reductase From A. Faecalis
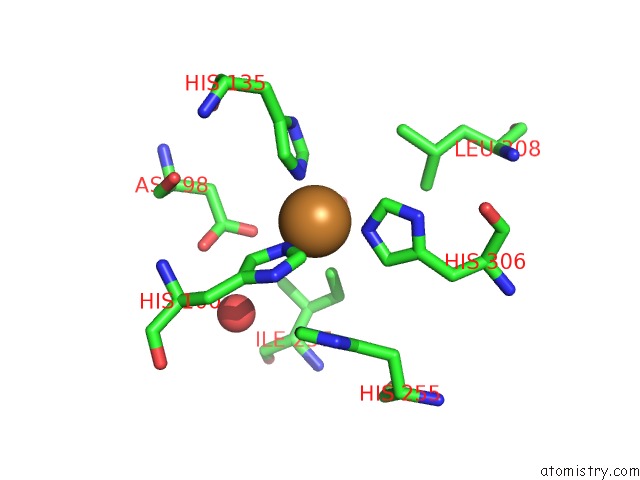
Mono view
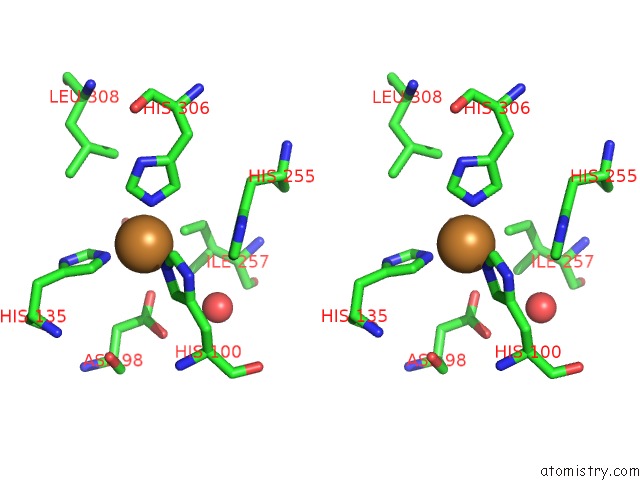
Stereo pair view

Mono view

Stereo pair view
A full contact list of Copper with other atoms in the Cu binding
site number 4 of Crystal Structure of Anaerobically Reduced Wild Type Nitrite Reductase From A. Faecalis within 5.0Å range:
|
Copper binding site 5 out of 6 in 2fjs
Go back to
Copper binding site 5 out
of 6 in the Crystal Structure of Anaerobically Reduced Wild Type Nitrite Reductase From A. Faecalis
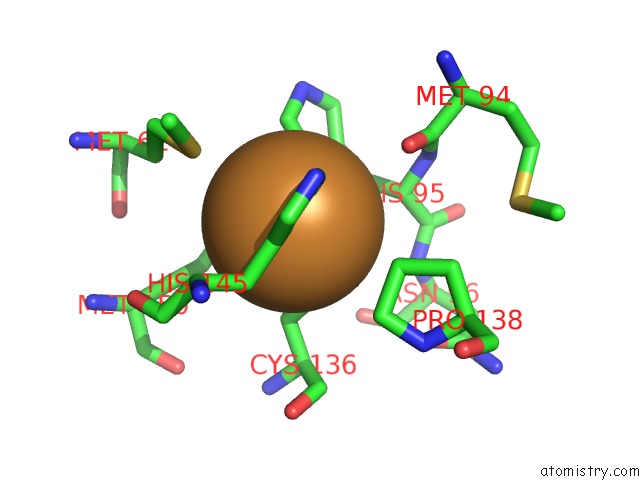
Mono view
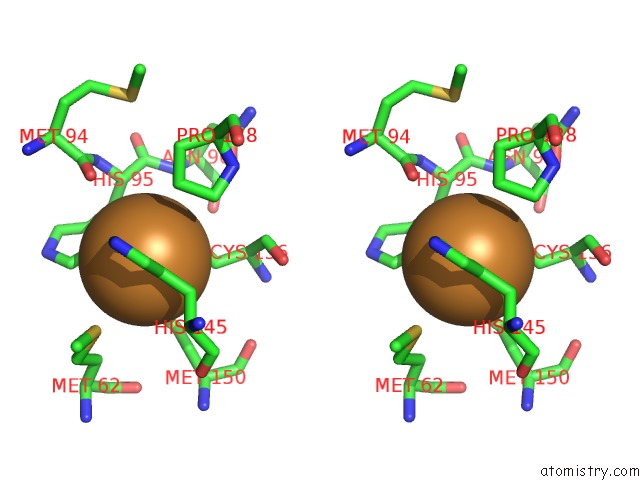
Stereo pair view

Mono view

Stereo pair view
A full contact list of Copper with other atoms in the Cu binding
site number 5 of Crystal Structure of Anaerobically Reduced Wild Type Nitrite Reductase From A. Faecalis within 5.0Å range:
|
Copper binding site 6 out of 6 in 2fjs
Go back to
Copper binding site 6 out
of 6 in the Crystal Structure of Anaerobically Reduced Wild Type Nitrite Reductase From A. Faecalis
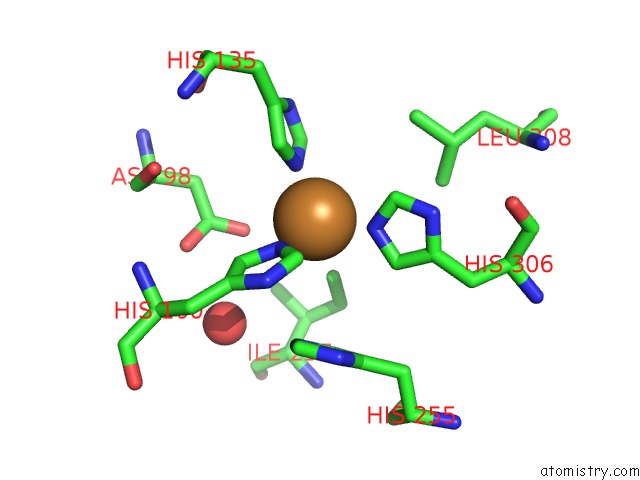
Mono view
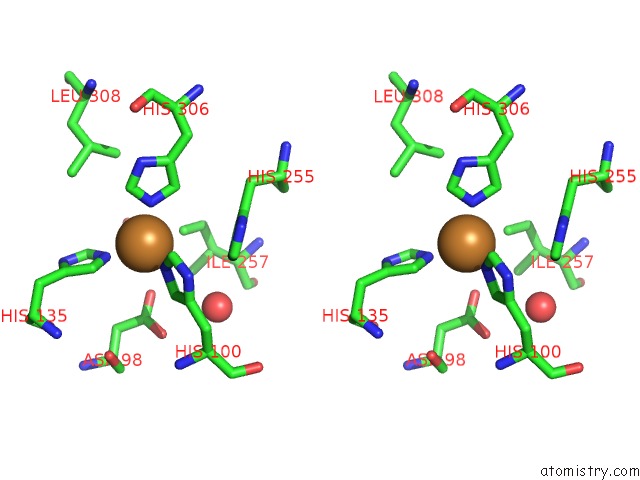
Stereo pair view

Mono view

Stereo pair view
A full contact list of Copper with other atoms in the Cu binding
site number 6 of Crystal Structure of Anaerobically Reduced Wild Type Nitrite Reductase From A. Faecalis within 5.0Å range:
|
Reference:
H.J.Wijma,
I.Macpherson,
O.Farver,
E.I.Tocheva,
I.Pecht,
M.P.Verbeet,
M.E.P.Murphy,
G.W.Canters.
Effect of the Methionine Ligand on the Reorganization Energy of the Type-1 Copper Site of Nitrite Reductase. J.Am.Chem.Soc. V. 129 519 2007.
ISSN: ISSN 0002-7863
PubMed: 17227014
DOI: 10.1021/JA064763J
Page generated: Tue Jul 30 23:29:21 2024
ISSN: ISSN 0002-7863
PubMed: 17227014
DOI: 10.1021/JA064763J
Last articles
Ca in 2P2KCa in 2P0R
Ca in 2P16
Ca in 2OX9
Ca in 2OYI
Ca in 2OZN
Ca in 2OZB
Ca in 2OY4
Ca in 2OYH
Ca in 2OY2