Copper »
PDB 1tmx-1x9l »
1ui8 »
Copper in PDB 1ui8: Site-Directed Mutagenesis of HIS592 Involved in Binding of Copper Ion in Arthrobacter Globiformis Amine Oxidase
Enzymatic activity of Site-Directed Mutagenesis of HIS592 Involved in Binding of Copper Ion in Arthrobacter Globiformis Amine Oxidase
All present enzymatic activity of Site-Directed Mutagenesis of HIS592 Involved in Binding of Copper Ion in Arthrobacter Globiformis Amine Oxidase:
1.4.3.6;
1.4.3.6;
Protein crystallography data
The structure of Site-Directed Mutagenesis of HIS592 Involved in Binding of Copper Ion in Arthrobacter Globiformis Amine Oxidase, PDB code: 1ui8
was solved by
H.Matsunami,
T.Okajima,
S.Hirota,
H.Yamaguchi,
H.Hori,
S.Kuroda,
K.Tanizawa,
with X-Ray Crystallography technique. A brief refinement statistics is given in the table below:
| Resolution Low / High (Å) | 10.00 / 1.80 |
| Space group | I 1 2 1 |
| Cell size a, b, c (Å), α, β, γ (°) | 157.762, 63.210, 184.244, 90.00, 111.64, 90.00 |
| R / Rfree (%) | 21.7 / 25.8 |
Copper Binding Sites:
The binding sites of Copper atom in the Site-Directed Mutagenesis of HIS592 Involved in Binding of Copper Ion in Arthrobacter Globiformis Amine Oxidase
(pdb code 1ui8). This binding sites where shown within
5.0 Angstroms radius around Copper atom.
In total 2 binding sites of Copper where determined in the Site-Directed Mutagenesis of HIS592 Involved in Binding of Copper Ion in Arthrobacter Globiformis Amine Oxidase, PDB code: 1ui8:
Jump to Copper binding site number: 1; 2;
In total 2 binding sites of Copper where determined in the Site-Directed Mutagenesis of HIS592 Involved in Binding of Copper Ion in Arthrobacter Globiformis Amine Oxidase, PDB code: 1ui8:
Jump to Copper binding site number: 1; 2;
Copper binding site 1 out of 2 in 1ui8
Go back to
Copper binding site 1 out
of 2 in the Site-Directed Mutagenesis of HIS592 Involved in Binding of Copper Ion in Arthrobacter Globiformis Amine Oxidase
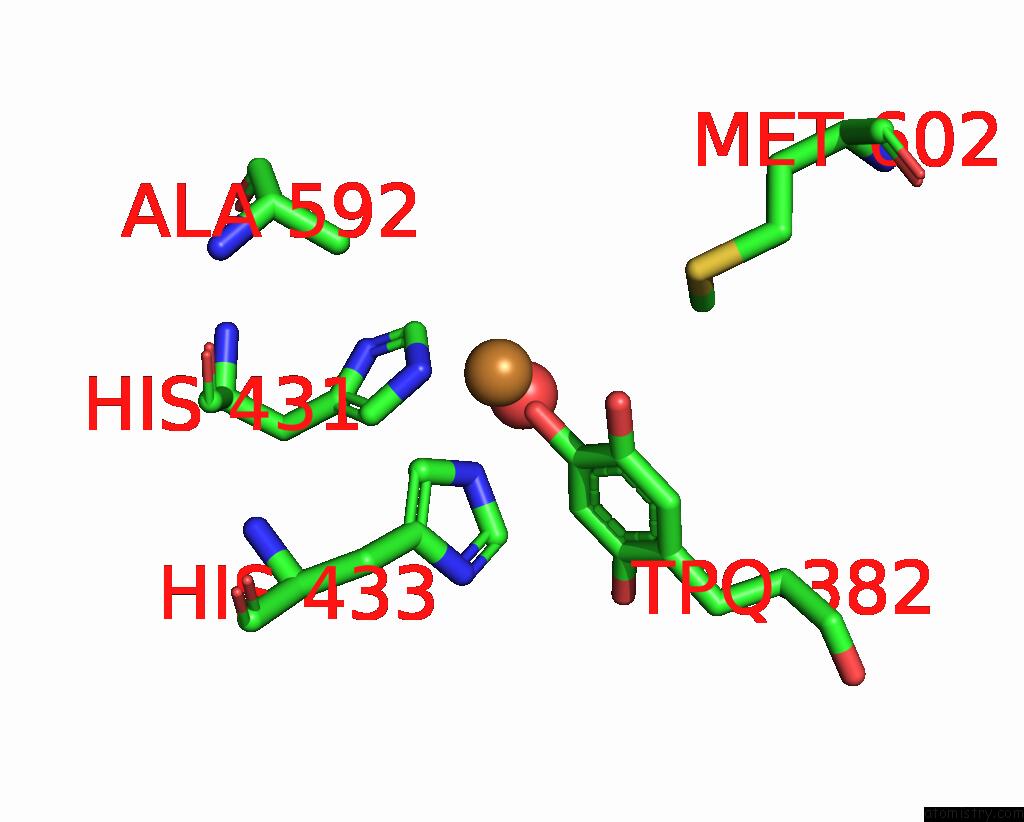
Mono view
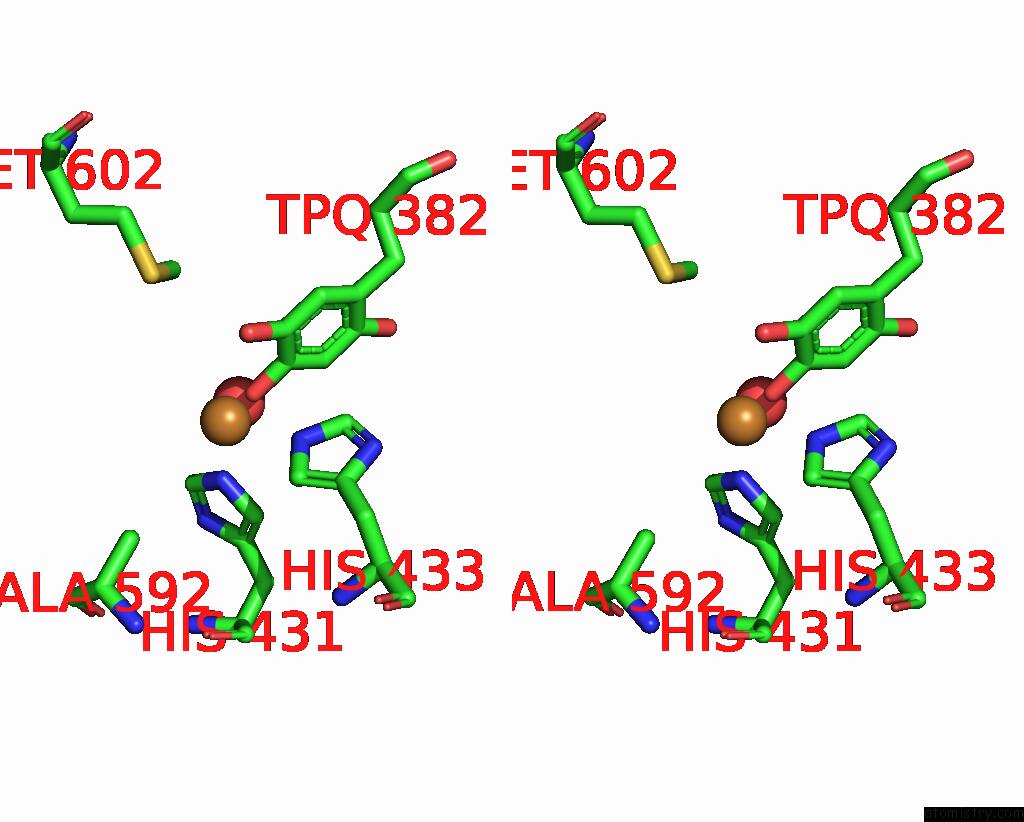
Stereo pair view

Mono view

Stereo pair view
A full contact list of Copper with other atoms in the Cu binding
site number 1 of Site-Directed Mutagenesis of HIS592 Involved in Binding of Copper Ion in Arthrobacter Globiformis Amine Oxidase within 5.0Å range:
|
Copper binding site 2 out of 2 in 1ui8
Go back to
Copper binding site 2 out
of 2 in the Site-Directed Mutagenesis of HIS592 Involved in Binding of Copper Ion in Arthrobacter Globiformis Amine Oxidase
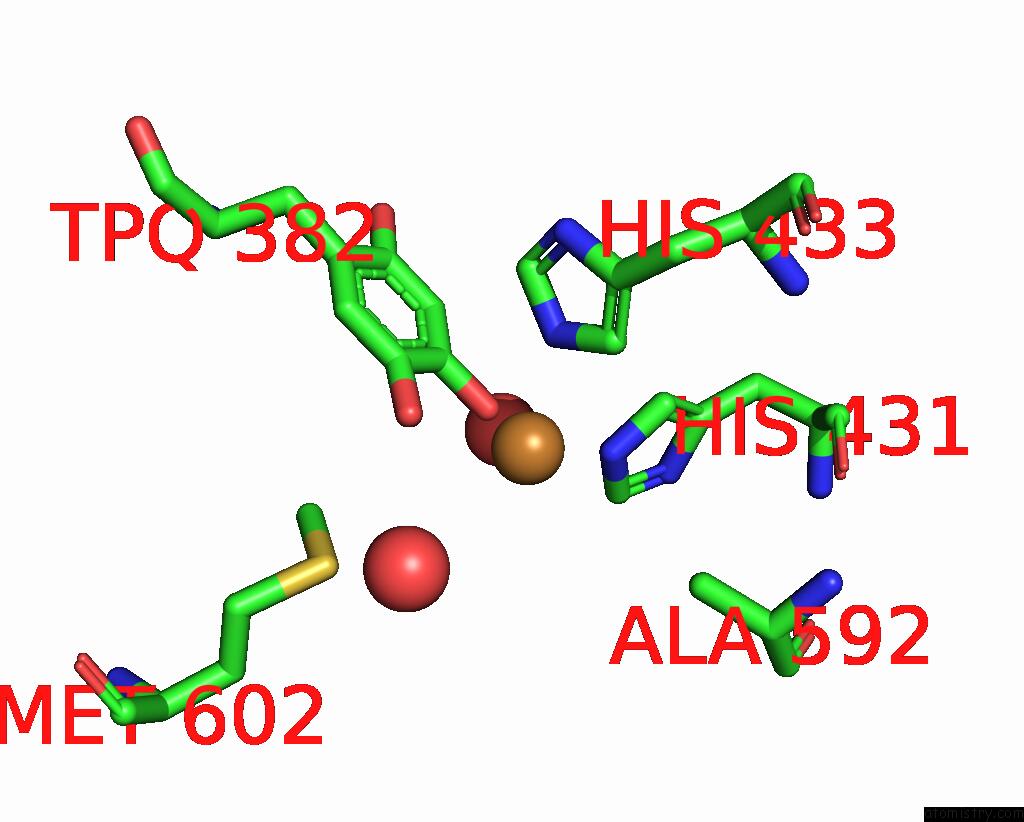
Mono view
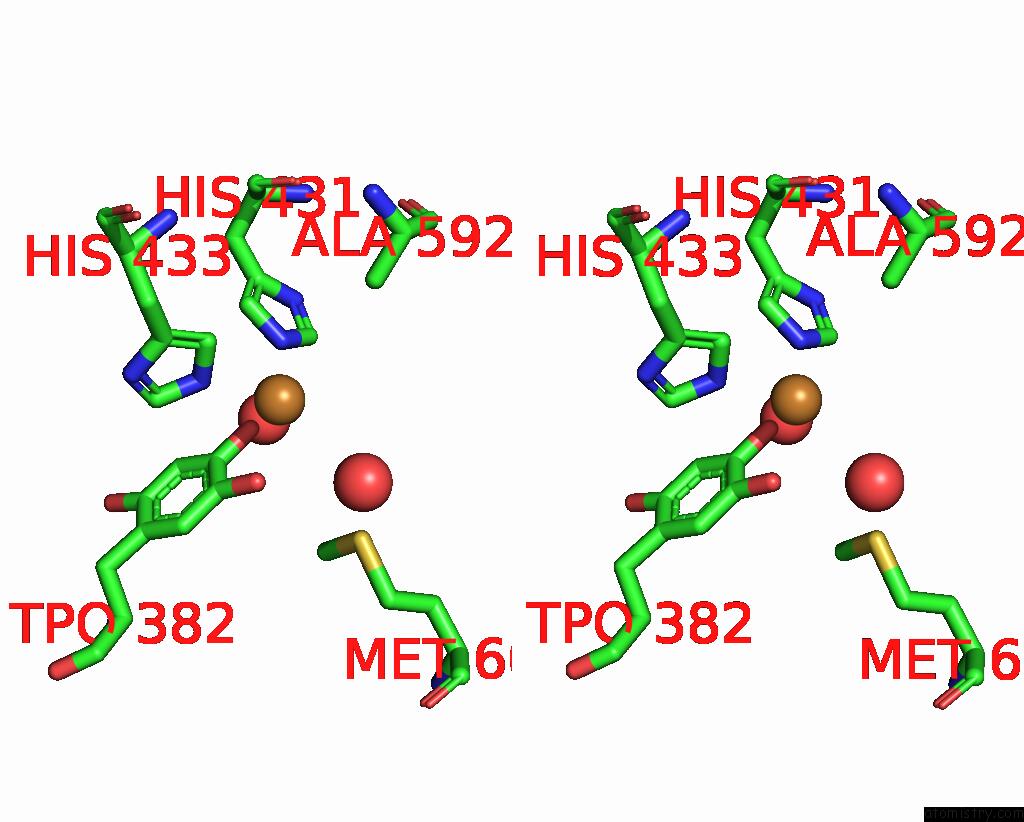
Stereo pair view

Mono view

Stereo pair view
A full contact list of Copper with other atoms in the Cu binding
site number 2 of Site-Directed Mutagenesis of HIS592 Involved in Binding of Copper Ion in Arthrobacter Globiformis Amine Oxidase within 5.0Å range:
|
Reference:
H.Matsunami,
T.Okajima,
S.Hirota,
H.Yamaguchi,
H.Hori,
S.Kuroda,
K.Tanizawa.
Chemical Rescue of A Site-Specific Mutant of Bacterial Copper Amine Oxidase For Generation of the Topa Quinone Cofactor Biochemistry V. 43 2178 2004.
ISSN: ISSN 0006-2960
PubMed: 14979714
DOI: 10.1021/BI0361923
Page generated: Mon Jul 14 00:29:56 2025
ISSN: ISSN 0006-2960
PubMed: 14979714
DOI: 10.1021/BI0361923
Last articles
Fe in 2YXOFe in 2YRS
Fe in 2YXC
Fe in 2YNM
Fe in 2YVJ
Fe in 2YP1
Fe in 2YU2
Fe in 2YU1
Fe in 2YQB
Fe in 2YOO