Copper »
PDB 1rju-1tl4 »
1rk5 »
Copper in PDB 1rk5: The D-Aminoacylase Mutant D366A in Complex with 100MM CUCL2
Enzymatic activity of The D-Aminoacylase Mutant D366A in Complex with 100MM CUCL2
All present enzymatic activity of The D-Aminoacylase Mutant D366A in Complex with 100MM CUCL2:
3.5.1.81;
3.5.1.81;
Protein crystallography data
The structure of The D-Aminoacylase Mutant D366A in Complex with 100MM CUCL2, PDB code: 1rk5
was solved by
W.L.Lai,
L.Y.Chou,
C.Y.Ting,
Y.C.Tsai,
S.H.Liaw,
with X-Ray Crystallography technique. A brief refinement statistics is given in the table below:
| Resolution Low / High (Å) | 29.40 / 1.80 |
| Space group | P 21 21 21 |
| Cell size a, b, c (Å), α, β, γ (°) | 60.016, 77.266, 135.982, 90.00, 90.00, 90.00 |
| R / Rfree (%) | 18.3 / 20.4 |
Other elements in 1rk5:
The structure of The D-Aminoacylase Mutant D366A in Complex with 100MM CUCL2 also contains other interesting chemical elements:
| Zinc | (Zn) | 1 atom |
Copper Binding Sites:
The binding sites of Copper atom in the The D-Aminoacylase Mutant D366A in Complex with 100MM CUCL2
(pdb code 1rk5). This binding sites where shown within
5.0 Angstroms radius around Copper atom.
In total only one binding site of Copper was determined in the The D-Aminoacylase Mutant D366A in Complex with 100MM CUCL2, PDB code: 1rk5:
In total only one binding site of Copper was determined in the The D-Aminoacylase Mutant D366A in Complex with 100MM CUCL2, PDB code: 1rk5:
Copper binding site 1 out of 1 in 1rk5
Go back to
Copper binding site 1 out
of 1 in the The D-Aminoacylase Mutant D366A in Complex with 100MM CUCL2
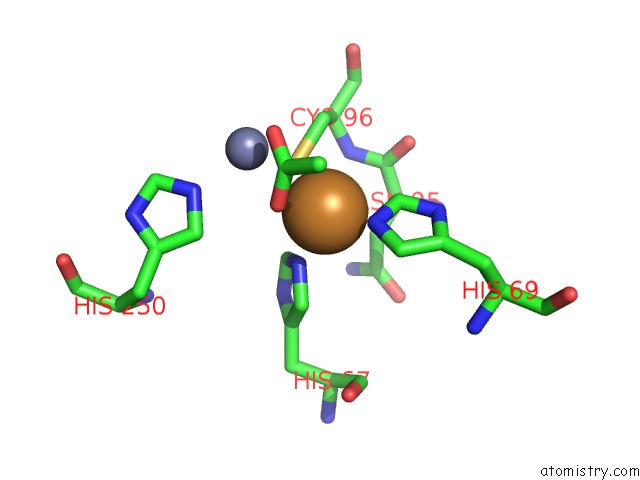
Mono view
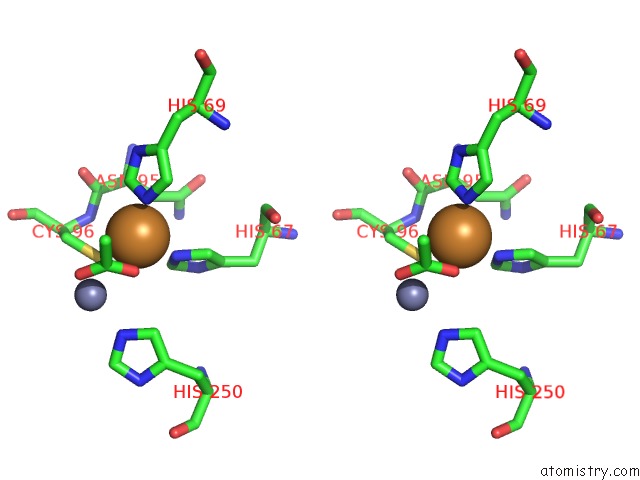
Stereo pair view

Mono view

Stereo pair view
A full contact list of Copper with other atoms in the Cu binding
site number 1 of The D-Aminoacylase Mutant D366A in Complex with 100MM CUCL2 within 5.0Å range:
|
Reference:
W.L.Lai,
L.Y.Chou,
C.Y.Ting,
R.Kirby,
Y.C.Tsai,
A.H.Wang,
S.H.Liaw.
The Functional Role of the Binuclear Metal Center in D-Aminoacylase: One-Metal Activation and Second-Metal Attenuation. J.Biol.Chem. V. 279 13962 2004.
ISSN: ISSN 0021-9258
PubMed: 14736882
DOI: 10.1074/JBC.M308849200
Page generated: Tue Jul 30 22:43:07 2024
ISSN: ISSN 0021-9258
PubMed: 14736882
DOI: 10.1074/JBC.M308849200
Last articles
Cl in 7U2ZCl in 7U31
Cl in 7U24
Cl in 7U1R
Cl in 7TZP
Cl in 7U0N
Cl in 7U1D
Cl in 7U00
Cl in 7U0O
Cl in 7U0A