Copper »
PDB 1eso-1haw »
1h1m »
Copper in PDB 1h1m: Crystal Structure of Quercetin 2,3-Dioxygenase Anaerobically Complexed with the Substrate Kaempferol
Enzymatic activity of Crystal Structure of Quercetin 2,3-Dioxygenase Anaerobically Complexed with the Substrate Kaempferol
All present enzymatic activity of Crystal Structure of Quercetin 2,3-Dioxygenase Anaerobically Complexed with the Substrate Kaempferol:
1.13.11.24;
1.13.11.24;
Protein crystallography data
The structure of Crystal Structure of Quercetin 2,3-Dioxygenase Anaerobically Complexed with the Substrate Kaempferol, PDB code: 1h1m
was solved by
R.A.Steiner,
B.W.Dijkstra,
with X-Ray Crystallography technique. A brief refinement statistics is given in the table below:
| Resolution Low / High (Å) | 49.39 / 1.90 |
| Space group | P 1 21 1 |
| Cell size a, b, c (Å), α, β, γ (°) | 109.272, 55.377, 123.929, 90.00, 98.33, 90.00 |
| R / Rfree (%) | 15.4 / 20.4 |
Copper Binding Sites:
The binding sites of Copper atom in the Crystal Structure of Quercetin 2,3-Dioxygenase Anaerobically Complexed with the Substrate Kaempferol
(pdb code 1h1m). This binding sites where shown within
5.0 Angstroms radius around Copper atom.
In total 4 binding sites of Copper where determined in the Crystal Structure of Quercetin 2,3-Dioxygenase Anaerobically Complexed with the Substrate Kaempferol, PDB code: 1h1m:
Jump to Copper binding site number: 1; 2; 3; 4;
In total 4 binding sites of Copper where determined in the Crystal Structure of Quercetin 2,3-Dioxygenase Anaerobically Complexed with the Substrate Kaempferol, PDB code: 1h1m:
Jump to Copper binding site number: 1; 2; 3; 4;
Copper binding site 1 out of 4 in 1h1m
Go back to
Copper binding site 1 out
of 4 in the Crystal Structure of Quercetin 2,3-Dioxygenase Anaerobically Complexed with the Substrate Kaempferol
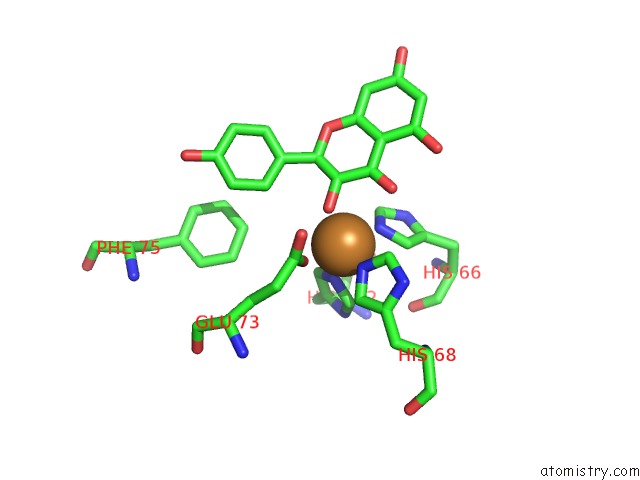
Mono view
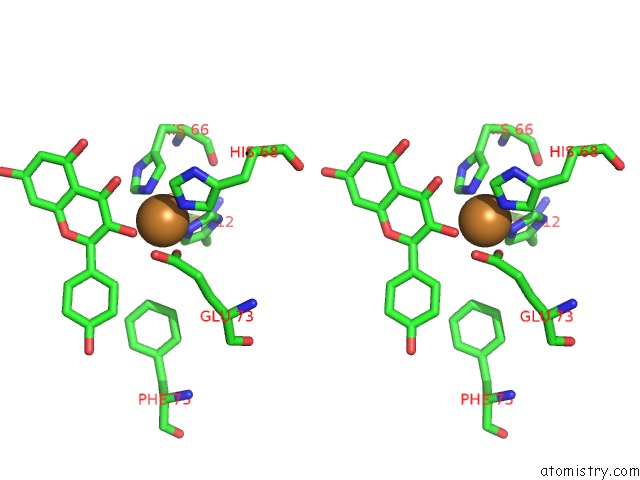
Stereo pair view

Mono view

Stereo pair view
A full contact list of Copper with other atoms in the Cu binding
site number 1 of Crystal Structure of Quercetin 2,3-Dioxygenase Anaerobically Complexed with the Substrate Kaempferol within 5.0Å range:
|
Copper binding site 2 out of 4 in 1h1m
Go back to
Copper binding site 2 out
of 4 in the Crystal Structure of Quercetin 2,3-Dioxygenase Anaerobically Complexed with the Substrate Kaempferol
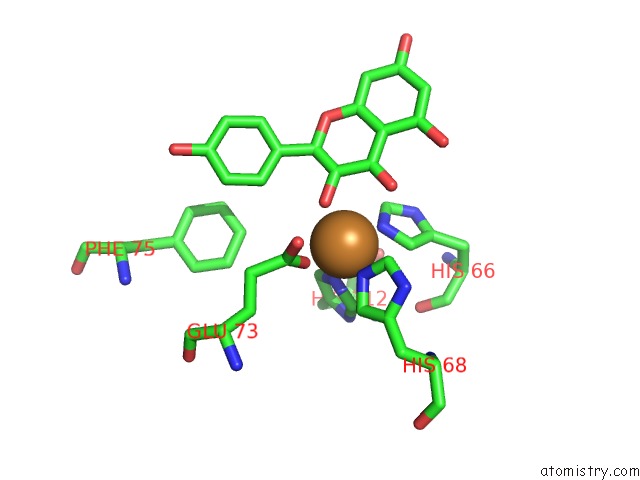
Mono view
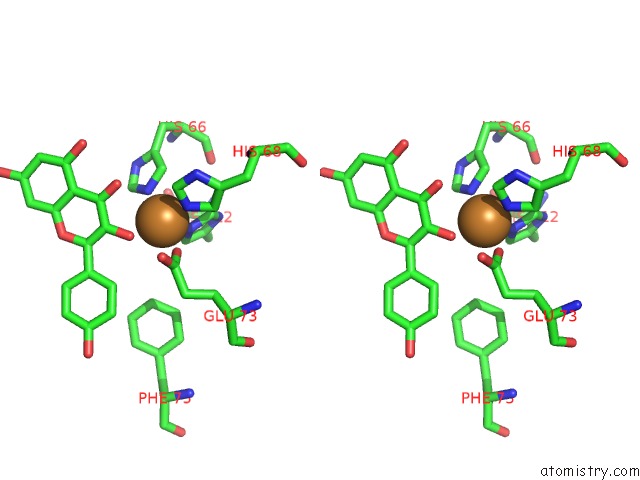
Stereo pair view

Mono view

Stereo pair view
A full contact list of Copper with other atoms in the Cu binding
site number 2 of Crystal Structure of Quercetin 2,3-Dioxygenase Anaerobically Complexed with the Substrate Kaempferol within 5.0Å range:
|
Copper binding site 3 out of 4 in 1h1m
Go back to
Copper binding site 3 out
of 4 in the Crystal Structure of Quercetin 2,3-Dioxygenase Anaerobically Complexed with the Substrate Kaempferol
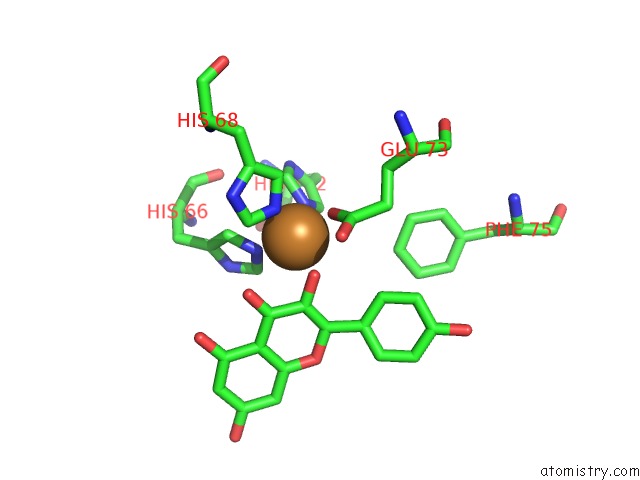
Mono view
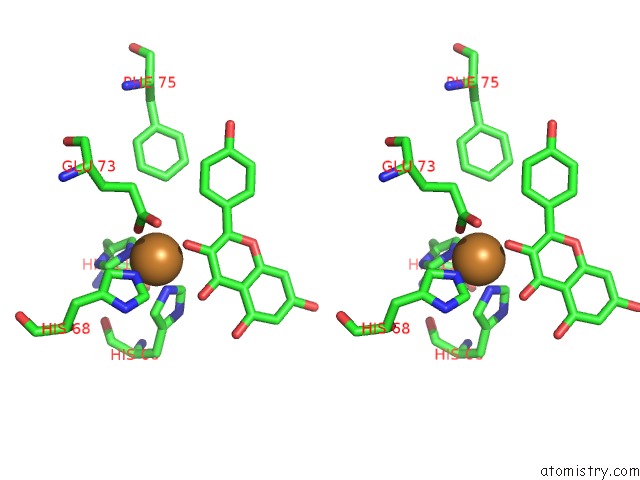
Stereo pair view

Mono view

Stereo pair view
A full contact list of Copper with other atoms in the Cu binding
site number 3 of Crystal Structure of Quercetin 2,3-Dioxygenase Anaerobically Complexed with the Substrate Kaempferol within 5.0Å range:
|
Copper binding site 4 out of 4 in 1h1m
Go back to
Copper binding site 4 out
of 4 in the Crystal Structure of Quercetin 2,3-Dioxygenase Anaerobically Complexed with the Substrate Kaempferol
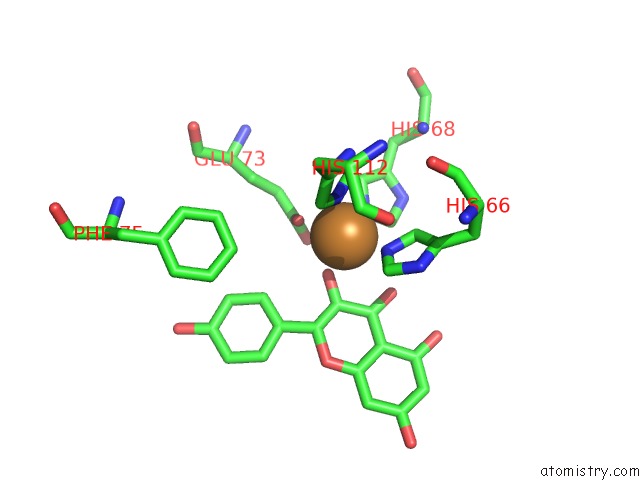
Mono view
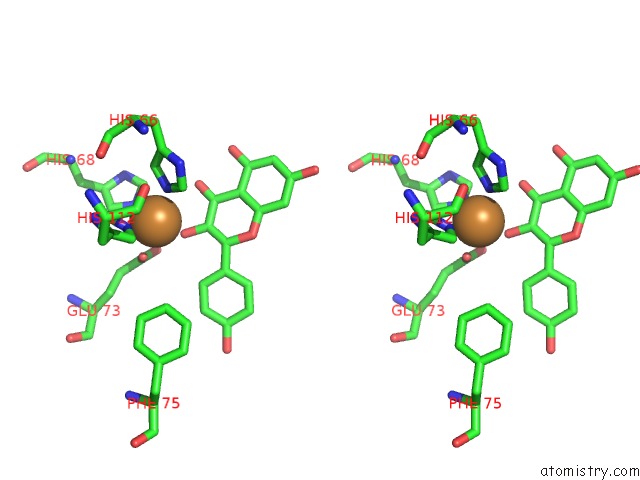
Stereo pair view

Mono view

Stereo pair view
A full contact list of Copper with other atoms in the Cu binding
site number 4 of Crystal Structure of Quercetin 2,3-Dioxygenase Anaerobically Complexed with the Substrate Kaempferol within 5.0Å range:
|
Reference:
R.A.Steiner,
K.H.Kalk,
B.W.Dijkstra.
Anaerobic Enzyme.Substrate Structures Provide Insight Into the Reaction Mechanism of the Copper- Dependent Quercetin 2,3-Dioxygenase. Proc.Natl.Acad.Sci.Usa V. 99 16625 2002.
ISSN: ISSN 0027-8424
PubMed: 12486225
DOI: 10.1073/PNAS.262506299
Page generated: Tue Jul 30 21:52:48 2024
ISSN: ISSN 0027-8424
PubMed: 12486225
DOI: 10.1073/PNAS.262506299
Last articles
Zn in 9JYWZn in 9IR4
Zn in 9IR3
Zn in 9GMX
Zn in 9GMW
Zn in 9JEJ
Zn in 9ERF
Zn in 9ERE
Zn in 9EGV
Zn in 9EGW